Q-Line Gasket
Q-line gaskets are the types of components used in applications, where sanitation is a priority. SSP Manufacturing, Inc. provides these sanitary gaskets in various specifications, and materials to suit your requirements. All our materials are FDA compliant.
Specifications of Q-Line Gaskets
We provide Q-line gaskets in the following specifications:

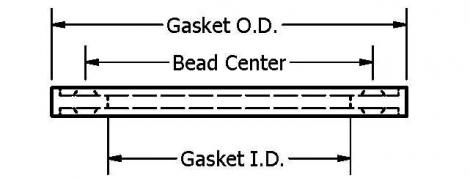
| Size |
Part# |
Gasket I.D. |
Gasket O.D. |
Bead Center |
| 1" |
40Q_-100 |
0.90 |
2.09 |
1.44 |
| 1-1/2" |
40Q_-150 |
1.40 |
2.12 |
1.70 |
| 2" |
40Q_-200 |
1.89 |
2.77 |
2.25 |
| 2-1/2" |
40Q_-250 |
2.37 |
3.46 |
2.85 |
| 3" |
40Q_-300 |
2.87 |
4.00 |
3.37 |
| 4" |
40Q_-400 |
3.87 |
5.00 |
4.40 |
Materials of Construction
Q-line sanitary gaskets available with us are made from the following materials:
- Buna N (40QH-XXX): Buna-N Q-line gaskets are well-known for their compression qualities, and flexibility. These gaskets are affordable, which makes them popular across various industries. Buna- N gaskets are not recommended for applications, where high temperatures are involved. These gaskets are perfect for the temperature range from -30°F to 250°F, and conforms to all FDA Title 21 CFR 177.2600, CGMP, and USDA, 3A® specifications. The Buna material does not pass U.S. Pharmacopeia Class VI Certification and Cytotoxicity.
- EPDM (40QE-XXX): EPDM Q-line gaskets offer great chemical resistance, and are suited for applications involving acetone, water, and ketone. These gaskets meet all FDA Title 21 CFR 177.2600, CGMP and USDA, 3A® specifications. EPDM gaskets are ideal for applications in the temperature range from -30°F to 400°F.
- FKM Fluoroelastomer (40QSFY-XXX): These gaskets are suitable for applications in the temperature range from -30°F to 400°F. FKM material offers excellent resistance against many alkalis and acids. These gaskets are not suitable for sterilization, and meets all FDA title USDA, 3A®, 21 CFR 177.2600, and CGMP specifications.
- PTFE (40IT-XXX):PTFE Q-line gaskets are ideal for applications, where low temperature flexibility is required. If used in large temperature variation applications, leakages may occur. These gaskets can resist process fluids, and have a low absorption rate. For continuous use, high pressure clamps should be used. These gaskets can be used in temperature ranges between -100°F and 500°F. PTFE material meets all the requisite CGMP, FDA Title 21 CFR 177.2600, and USDA, 3A® specifications, and USP Class IV, EP 3.1.9, and cytotoxicity specifications.
- Silicone (40IX-XXX):Silicone Q-line gaskets are largely used in many pharmaceutical industries, due to their non-leechable characteristics. These gaskets are ideal for a wide temperature range from -40°F to 450°F. Our silicone sanitary Q-line gaskets can be autoclaved or hand cleaned, which makes them popular across various food processing units, too. These gaskets meet all FDA Title 21 CFR 177.2600, CGMP, and USDA, 3A® specifications.
- Tuf-Steel® (40IT-TS-XXX):Tuf-Steel® gaskets are designed to meet stringent sanitary requirements in many industries. The Tuf-Steel material helps withstand extreme heat and various harmful chemicals. Also, it is a non-stick material with properties like ultra-low absorption, zero pigmentation, and minimum creep, as well as cold flow. These properties help the gaskets ensure leak-free operations. These Q-line gaskets can be used for steam and water applications for extended period of time. These gaskets meet all required BPE, ASME, and USDA 3A, and CGMP specifications. The gaskets can be used in the temperature range from -320°F to 550°F.
Industries Using Q-Line gaskets
Our Q-line gaskets are used across the following industries:
- Food and beverage processing
- Biotech
- Pharmaceutical
- Semiconductor
- Dairy
- Water for Injection (WFI)
- Ultra-pure water processing
- Ice cream processing units
SSP Manufacturing, Inc. is one of the trusted names in seals and sanitary gaskets used in various industries. With an industry experience spanning over two decades, we are quick to understand requirements in diverse industries. For any queries regarding sanitary gaskets provided by us, please call us on 888-238-7325 or send us an email at rrom@sspseals.com.
Quality Assurance Documentation
SSP Manufacturing Inc ships all products with a certificate of conformance.
It is SSP Manufacturing Inc policy to provide the highest quality products, which consistently meet the product specifications developed by SSP Manufacturing Inc and their customers, both internal and external. We are committed to the continuous improvement of our quality system. We will meet and exceed the expectations of our customers.
Gaskets Meet the Most Stringent Standards For Purity:
- U.S. Pharmacopeia Class VI Certification*
- Cytotoxicity Criteria*
- Title 21CFR177.2600 and 177.1550
- Current Good Manufacturing Practices (CGMP)
- ASME-BPE Standards
- ISO9001:2000 and QS-9000:1998
- Animal Derived Ingredient Free*
*Buna does not pass U.S. Pharmacopeia Class VI Certification And Cytotoxicity and is not ADI Free®.
 sales: gm@sspseals.com Toll Free: +1-888-238-SEAL Request A Quote
sales: gm@sspseals.com Toll Free: +1-888-238-SEAL Request A Quote


